Coppertone is voluntarily recalling five spray-on, SPF 50 sunscreen brands after it detected benzene, a known cancer agent, in them.
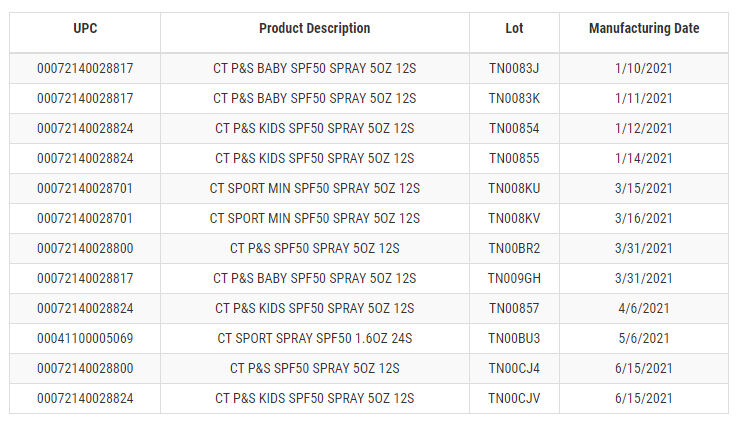
The voluntary recall is for three types of Coppertone Pure & Simple SPF 50 Spray, Coppertone Sport Mineral SPF 50 Spray and Travel-Size Coppertone Sport Spray SPF 50, all of which were manufactured between Jan. 10, 2021 and June 15, 2021.

Two of the aerosol spray sunscreens were marketed for use on infants and children.
You can see the full list of batches that were recalled, and when they were manufactured, on the Food and Drug Administration’s website.
The FDA says benzene is classified as a human carcinogen. Exposure either orally, through inhalation or on skin, can result in an increased risk of cancers, such as leukemia, blood cancer of the bone marrow and other life-threatening blood disorders, the FDA says.
In its announcement on the FDA’s website, company said, “Daily exposure to benzene at the levels detected in these affected Coppertone aerosol sunscreen spray products would not be expected to cause adverse health consequences based on generally accepted exposure modeling by numerous regulatory agencies.”
Coppertone said it has alerted retailers about the recall and is arranging for returns of all voluntarily recalled lots of sunscreen spray products.
Adverse reactions or quality problems experienced with the use of Coppertone’s products can be reported to the FDA’s MedWatch Adverse Event Reporting program.
Anyone who has experienced problems that may be related to using Coppertone’s recalled products should get in touch with a physician or a health care provider.
The company is offering refunds.
Other major sunscreen manufacturers have recalled their own products after detecting traces of benzene in them.
Johnson & Johnson recalled five of its spray-on sunscreen products in July after it found low levels of the chemical.
Online pharmacy and lab Valisure found benzene in Banana Boat, CVS and Neutrogena branded lotions and spray-on products back in June.