The US has an opportunity to get ahead in the Covid-19 pandemic by getting more people vaccinated — and three key things can help address ongoing concerns, one expert says.
“People were worried about safety, we now have hundreds of millions of doses out there, so we have great data on safety,” emergency medical physician Dr. Anand Swaminathan told CNN on Saturday. “People were worried about efficacy,” he said, adding there is now real-world data showing how effective the vaccines are.
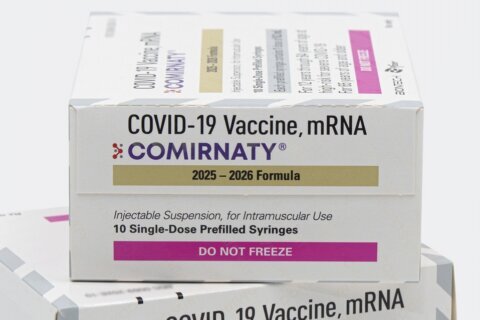
And finally, Swaminathan said, some Americans were concerned that the vaccines didn’t have approval from the Food and Drug Administration and had only received emergency use authorization. But Pfizer/BioNTech announced Friday they have initiated an application to the agency for full FDA approval of their vaccine for people 16 and older — making it the first Covid-19 vaccine in the US to be assessed for full approval from the FDA.
“What’s the difference between FDA emergency use authorization and full approval? It’s really time and money,” infectious disease specialist and epidemiologist Dr. Céline Gounder told CNN on Saturday.
“But for some people, seeing a full approval from the FDA will indeed give them more confidence that these vaccines are safe and effective — and look, they are safe and effective,” she added.
The FDA “will move as expeditiously as possible,” without compromising its safety standards, to assess Pfizer’s Covid-19 vaccine for approval, White House Covid-19 response coordinator Jeff Zients said Friday.
More than 151 million Americans — roughly 45.6% of the US population — have received at least one Covid-19 vaccine dose, according to data from the Centers for Disease Control and Prevention.
More than 112 million Americans — nearly 34% of the population — are fully vaccinated, CDC data shows.
US official: Vaccine confidence only one piece of puzzle
For officials across the country, getting more Americans vaccinated will now be an uphill battle, as experts say the US has reached the crowds who weren’t as eager to get a shot or still have questions.
Vaccination rates are already falling. For the first time since early March, the seven-day average of Covid-19 vaccine doses administered in the US fell below 2 million per day, according to CDC data published on Saturday.
But confidence in the vaccines is only “one piece of the puzzle,” when it comes to the challenges the US faces in its vaccination efforts, US Surgeon General Dr. Vivek Murthy said during a White House Covid-19 briefing on Friday.
“The barriers to getting vaccinated fall into three main categories,” he said. “Vaccine confidence, motivation and access.”
And local, state and federal efforts are ongoing, he said, which aim to address all three pieces.
“I know it’s been a difficult year and that everyone in our country has been asked to step up and sacrifice in a big way,” Murthy said. “I want to be clear that this pandemic will end. The faster we get vaccinated, the faster that day will come.”
Last week, President Joe Biden announced a new goal of administering at least one Covid-19 vaccine dose to 70% of US adults by July 4. As of Saturday, four states have already done so: Connecticut, Hawaii, Massachusetts and Vermont.
J&J pause ‘cast a shadow’ on vaccinations
One factor that may have contributed to the slowing vaccinations was the recommended pause on the Johnson & Johnson Covid-19 vaccine, one expert said Saturday.
That recommendation was lifted last month and officials said the label would be updated to warn of blood clot risks. Experts concluded the vaccine’s benefits outweighed its “known and potential risks” and the vaccine continued to meet “standards for safety, effectiveness and quality,” acting FDA Commissioner Dr. Janet Woodcock said at the time.
But the recommendation to pause had its own effects.
“(The) Johnson & Johnson pause did cast a shadow over the momentum that we had gained,” Dr. Jayne Morgan, clinical director of the Piedmont Healthcare Covid Task Force, said. “I think people who were on the fence climbed down from the fence and once again are peeking over as we move toward what we hope will be a Pfizer full FDA approval.”
Two weeks after the J&J recommended pause was lifted, that vaccine accounts for a very small share of doses administered in the US — and the current pace of administration lags significantly from the pace before the pause, CDC data shows.
Over seven days, the J&J vaccine accounted for just about 3.5% of total doses administered, according to data published Friday by the CDC.
Boosters will likely be a reality, expert says
Meanwhile, the CDC is preparing for seasonal Covid-19 vaccine boosters in case they are needed, its chief said Friday, adding researchers are also looking into whether the US will need a booster that specifically works against certain coronavirus variants.
“We are doing the studies on boosters to see if we will need them,” CDC Director Dr. Rochelle Walensky told actress Jennifer Garner in an interview streamed on Instagram. “We want to be prepared for them should we need them.”
Potential Covid-19 boosters could be offered in a similar way to the annual flu vaccine, Walensky said.
“We hope we don’t have to do it every season, but we’re preparing in case we do,” she added.
A batch of new studies published last week show how well Covid-19 vaccines work against worrying new variants — and one indicates booster doses can help them work even better.
“I think most likely, we are going to be recommending boosters,” Swaminathan told CNN Saturday. “Whether that be at six months, at a year, we still don’t know. What we do know, is that even against the variants, our vaccines are still pretty effective.”
“But I think boosters are going to be a reality,” he added.