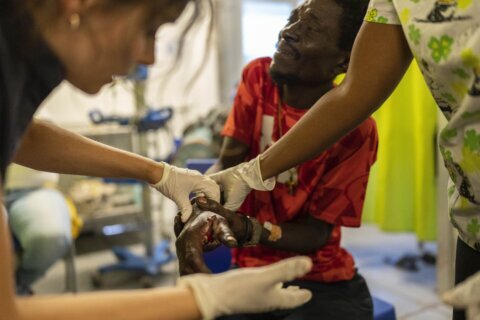
When Steve Lagoudis was diagnosed with bladder cancer two years ago, his doctor gave him a powerful immunotherapy that was the standard of care: bacillus Calmette-Guerin, a decades-old drug originally developed as a vaccine against tuberculosis. The initial treatment didn’t work, but when he sought a second opinion from a specialist at a cancer hospital near his home in Great Neck, New York, the urologist recommended another round of BCG. Unfortunately for Lagoudis, the hospital didn’t have the lifesaving medication in stock because of severe shortages. “I was shocked,” says the 60-year-old banker. “You hear things like this in developing countries but not in the U.S. It’s outrageous.”
Without this treatment, Lagoudis’ only alternative was a radical surgery to remove his bladder, which would mean wearing a urine collection bag for the rest of his life. But he finally landed at Mount Sinai Hospital in Manhattan, which did have BCG. The additional treatment, combined with an experimental drug, did in fact eradicate his cancer. He now receives a maintenance dose of BCG every three months to prevent a recurrence. “I was lucky,” says Lagoudis, who nevertheless flew to his native Greece to purchase backup supplies of BCG. “I want to make sure I have it.”
[Read: Types of Bladder Cancer]
Only one company, Merck, makes BCG here in the U.S., and the company is unable to keep up with demand. ” Bladder cancer can be lethal but if caught early, the majority of patients can stop progressing,” says Ashutosh Tewari, system chair of the department of urology at the Icahn School of Medicine at Mount Sinai Health System. “BCG can not only cure but preserve the bladder, which preserves their quality of life. But we’ve had to prioritize people” — only giving it to those who could benefit the most — “to make sure there is enough.”
Drug shortages like this have become so common that in 2018 nearly 200 occurred in hospitals nationwide, according to the American Society of Health-System Pharmacists; more are expected when tallies are completed for 2019. Many medical associations have raised alarms about the dire consequences these shortages pose, which include unsafe practices, compromised patient care and potentially harmful medication errors.
Between January 2008 and March 2014, in fact, inadequate supplies of lifesaving therapies quadrupled and shortages of drugs for which there is no alternative more than doubled, according to an analysis published in Academic Emergency Medicine. “The shortages have become our new reality,” says Stephanie Hall, chief medical officer of Medical Center of USC and USC Norris Comprehensive Cancer Center in Los Angeles. They’re “often difficult to predict. Sometimes, we only get wind of a shortage when we try to purchase a medication.”
And in 2020, the COVID-19 pandemic only exacerbated the problem. The sudden tidal wave of severely ill patients placed unprecedented strains on the U.S. health system, triggering a scarcity of many vital medications due to the unanticipated spike in demand. The pandemic also spotlighted serious flaws in the global supply chain, because many finished medications, bulk ingredients or raw materials are produced in countries hard hit by COVID-19, like China and India. “There are aspects of our supply that are supplied almost 100% from other countries,” says Kavita Patel, a Washington, D.C., physician who served as a policy director in the Obama White House and is a nonresident fellow at the Brookings Institution.
The Cost Factor
Several other factors are fueling this crisis. Some older drugs are less expensive, prompting drugmakers to halt production because of slim profit margins. The chemotherapy vincristine, the mainstay of treatment for many childhood cancers and critical for youngsters suffering from acute lymphoblastic leukemia, was only made by two companies before July of 2019, when Teva Pharmaceutical Industries abandoned the market and sparked a shortage. “These are all economic and business decisions, and it’s a persistent and pervasive problem,” says Yoram Unguru, a pediatric hematologist/oncologist at The Herman & Walter Samuelson Children’s Hospital at Sinai and a core faculty member at Johns Hopkins Berman Institute of Bioethics in Baltimore. “We’re not seeing shortages of blockbuster drugs that make millions of dollars.”
Regulatory changes can also prompt drugmakers to stop making older drugs that were grandfathered in before the Food and Drug Administration required safety and efficacy data, because they have no financial incentives to fund new tests. Plus, facility upgrades to meet FDA standards “have forced plants to close for extended periods,” says Mark Todd, chief pharmacy officer at University of Alabama at Birmingham Hospital. “Sometimes, there may be only one or two manufacturers to pick up the extra demand.”
Mitigating the Impact
But the biggest culprits, accounting for nearly two-thirds of shortages, are quality concerns both here in the U.S. and arising from poor manufacturing standards in countries like China and India that lead to contaminated drugs entering the market. That’s the finding of a 2019 FDA analysis that also noted that shortages are increasing and lasting longer in what the agency called “a broken marketplace.” Since 2013, pharmaceutical companies based in the U.S. and abroad have recalled about 8,000 medicines.
Natural disasters can cause shortages, too. In 2017, when Hurricane Maria ravaged Puerto Rico, home to nearly 50 major drug and medical supply makers, the storm knocked out dozens of facilities, leading to shortages of medications and IV saline bags. For months afterwards, hospitals nationwide were in crisis mode, and nurses had to resort to standing at patient bedsides slowly injecting medications like IV antibiotics by syringe instead of letting the medication drip in from an IV bag. Pharmacists have formulated guidelines for managing drug shortages that “can help mitigate some of the impact,” says Erin Fox, senior director of drug information and support services at the University of Utah Health system, who is leading this effort. “But it takes time and valuable resources.”
In some urgent situations, hospitals turn to neighboring facilities for help. When Hurricane Maria hit, Boston Medical Center didn’t have enough plastic tubing in stock for the repeater pumps that it was using to prepare vials of antibiotics. “We reached out to one of our local hospitals,” says Joy Vreeland, senior director/associate inpatient pharmacy at BMC in Boston, “and they had plenty of extra tubing that helped carry us through the crisis.”
Similarly, Keck Hospital of USC has established cooperative relationships with other facilities in Los Angeles. “Some hospitals refer patients to us because they can’t get certain drugs and vice versa,” Hall says.
[READ: Do Drug Company Payments to Doctors Influence Which Drugs They Prescribe?]
A New Strain
The COVID-19 pandemic worsened global disruption of the supply chain, and that exacerbated shortages of nearly 40% of the drugs (including epinephrine, the asthma inhaler albuterol and certain antibiotics) considered essential for critical care, according to a March 2020 analysis by University of Minnesota researchers. “Just in time” manufacturing practices, which drugmakers use to avoid being left with unused inventory, worsened the problem because there was no slack in the system when demand rose suddenly. As a result, the unexpected surge in gravely ill COVID-19 patients suffering from respiratory or kidney failure caused severe shortages of medications such as fluids, sedatives, antibiotics, paralytic agents, fentanyl and other supplies needed when patients require emergency dialysis or to be put on ventilators.
In response to the crisis, says the University of Utah’s Fox, “drugmakers are ramping up production as fast as they can, and hospitals across the country are trying to buy enough to be prepared and stocking up on second- and third-line medications in case they have to use some of these older drugs.”
Meanwhile, the initial absence of effective treatments for COVID-19 has prompted a push to repurpose already approved medications. Scientists have raced to screen thousands of drugs to identify candidates that might work against the virus. If a drug has already gone through safety and efficacy testing for FDA approval for one condition, it can skip some steps if being considered for another use. “The path from an experiment in the test tube to a clinical trial is much faster if you’re starting with an existing drug than if you have to build one from scratch,” says Todd Golub, chief scientific officer and director of the cancer program at the Broad Institute in Cambridge, Massachusetts. It also lowers the overall development costs.
Repurposing has a long history. The cancer drug AZT was the first medication approved to combat AIDS, while the antipsychotic medication chlorpromazine (thorazine) has been used to treat allergies, arthritis and even irregular menstrual periods.
In a similar vein, drugs that combat other virally caused illnesses, like hepatitis C or HIV, or that have shown efficacy against other syndromes caused by coronaviruses, like SARS or MERS, might be found to keep COVID-19 in check, too. Initially, the anti-malaria drugs hydroxychloroquine and chloroquine attracted widespread attention when a small but controversial French study suggested they might combat COVID-19, but the intense publicity and high-profile advocates sparked unintended consequences. Even though these drugs can cause severe adverse reactions and no large-scale human tests had proven their efficacy against the coronavirus, the federal government stockpiled millions of doses of the medications. (The FDA revoked its emergency use authorization for oral formulations of the drugs for COVID-19 in June based on adverse clinical data.) There was also a run on the drugs by doctors and other purchasers. As a result, some patients with lupus and other autoimmune diseases like rheumatoid arthritis who depend on chloroquine for its approved uses — to treat their illnesses — were forced to ration their pills or contact a dozen or more pharmacies to get their prescriptions filled.
Despite these setbacks, research continues on other potential COVID-19 therapies, like remdesivir, first formulated to treat Ebola, which was given emergency use authorization by the FDA for those with “severe COVID-19” in May. “We’re targeting a number of different proteins the virus needs for replication,” says Karla Satchell, a microbiologist at Northwestern University Feinberg School of Medicine in Chicago and a lead scientist for a team helping develop drugs to combat COVID-19. “We’re screening all of these drugs for their ability to stop the virus.”
Do-it-Yourself
Some states now also allow compounding pharmacies to make their own medications — another strategy for alleviating shortages. While many hospitals have always compounded products, the feds created a new category of pharmacies, called 503B, which may (after registering with the FDA and subject to state oversight) produce for other health care facilities large batches of product that are unavailable through regular channels. “Think diluting a medication into a syringe so it’s ready for an anesthesiologist,” says Fox. “Commercial companies don’t sell these, so there is a demand to compound them.”
Major hospital systems like Cleveland Clinic and Mayo Clinic have licensed pharmacies in place throughout their facilities doing compounding. Vanderbilt University Medical Center in Nashville is now outsourcing to 503B pharmacy compounders but plans to build compounding capacity in-house to reduce its reliance on traditional drugmakers. “We actually compound a lot of things, like opiates for pain and IV morphine for surgery,” says Scott Knoer, Cleveland Clinic’s chief pharmacy officer. “We’re fortunate here because not every hospital can do this.”
The shortages have gotten so bad that a consortium of health care systems have banded together to create a nonprofit company to manufacture generic medications. Called Civica Rx, the nonprofit contracts with drugmakers to make generics that are in short supply. Fourteen drugs, including the blood thinner heparin and dexamethasone, a steroid, are already available, and more are on the way. “This should help in the future,” says Fox, who is on Civica’s advisory board. “By the end of this year, they should have about 90 products.”
Switching Medications
To avoid interruptions in patient care, pharmacists track possible shortages and seek to stock alternatives that are as effective. But these substitutes are often many times more expensive — normally, it’s the older, cheaper drugs that are in short supply — and there may be problems with suppliers they haven’t used before. “We actually developed a dashboard here at the Clinic to combine all the data associated with drug recalls and drug inspections so we’re not buying products from a facility that hasn’t performed well,” says Jeff Rosner, senior director of pharmacy sourcing and purchasing at Cleveland Clinic. “We don’t want to substitute a drug that is having problems with the FDA.”
And being forced to turn to alternatives creates another set of headaches for providers and health care institutions. “Switching out products and using alternatives is time-consuming for a host of reasons,” Fox says. “You have to meet with clinicians to make sure the new drugs are appropriate, educate the nursing staff, and change the automation, including bar codes, scanners, smart pumps and the electronic medical record itself. All of this has to be changed back when the shortage ends.”
[Read: How to Get All Your Medications Without Going to the Pharmacy.]
Out of Options
Sometimes there are no alternatives, which can put patients at grave risk. Heparin, for instance, is in short supply, and cardiac surgeons worry about doing open heart surgery without this crucial blood-thinning medication, which has been proven to prevent stroke-causing clots from forming during lengthy, complex procedures; other blood thinners don’t have enough scientific evidence that show they work as well.
There are also serious shortages of immunoglobulin, or IVIG, that is used to treat “a huge variety of diseases, including immune disorders like lupus and multiple sclerosis, and bone marrow transplants, and there really aren’t therapeutic alternatives,” says Gina Caliendo, director of clinical pharmacy service at Mount Sinai Hospital. Shortages are triggered by problems in the supply chain. Since the drug is derived from the blood plasma of thousands of donors, it can take anywhere from eight to 10 months to ramp up production if there is increased demand.
For parents like Sonia Green, the stress and uncertainty is personal. Three of her sons, who range in age from 15 to 21, suffer from primary immune deficiency disease. This genetic condition interferes with the normal functioning of immune system cells called B-cells, which produce a protein needed to fight off infections. IVIG rectifies this by supplying healthy antibodies that can fight disease. Monthly infusions enable her children to live a normal life rather than being home-schooled — two of the brothers attend high school while her oldest son is a senior at the University of Pittsburgh. “I’m always holding my breath, and it feels like we’re on a tightrope every month,” says Green, a law professor in Chicago. “If they couldn’t get IVIG, it would completely upend their lives.”
If a patient is dependent on lifesaving therapies, it’s important that he or she stay in communication with physicians to ensure continuity of care when treatment needs to be delayed or changed, advises Fox. “Sometimes another treatment can be just as effective, and it will be available.”
More from U.S. News
5 Common Preventable Medical Errors
9 Strategies for Reducing Emergency Room Medication Errors
8 Medications That Treat Multiple Conditions
Tackling Dangerous Drug Shortages originally appeared on usnews.com